Outreach
As a group we undertake a range of outreach activities. For more information on our outreach activities relating to the science of a jet engine, see our related page at: https://www.rrutc.msm.cam.ac.uk/outreach/.
What do we do?
Here in the Structural Materials Group, we are all Materials Scientists. Check out the sections below to find out more about what we do.
Materials Science is all about understanding:
- how things are made, whether by nature or changed by us,
- how they can be used, and
- how they can be made to perform better for ever more demanding uses.
Materials Science fits in the space between engineering, physics and chemistry, combining the design of innovative machines and devices with an understanding of the rules and limits of the natural world. A surprisingly large number of important scientific and engineering problems in today’s world are based on a lack of suitable materials; this might be because better materials are too expensive or dangerous to obtain, or we simply do not have enough research into alternatives. This means that breakthroughs in Materials Science can have a big impact on how technology evolves.
Materials are what everything is made of and can be natural, like wood, or designed and made by humans, like plastic – in total, the list is very long with about 300 000 different known materials.
A big step in the field was the Space Race; in the past century this has helped define the basis of much of Materials Science research, with metals and their alloys for the hull, silicon for the electronics and carbon for some of the wackier applications like heat shields for re-entry.
Today, research goes much further than the original trio of metals, ceramics and polymers to now include nanotechnology and biomaterials, which between them focus on creating materials with novel structures and unique functionalities from both organic and inorganic building blocks down to the nanometre scale. This is often inspired by nature itself – think about how strong and light bones are; so, by using the design of a bird’s wings, we can make lighter and more fuel efficient aircraft.

A long standing member of the Materials Science world is Metallurgy. It is to a blacksmith what medical science is to a medic: it goes beyond mystery and folklore, providing us with the reasons and understanding. Metallurgy is one of the oldest forms of Materials Science and has been key in human development, literally defining the ages: Stone, Bronze and Iron Ages. Nowadays it remains a thriving scientific field that studies the behaviour and properties of metals to improve manufacturing and create a better product.
The role of the metallurgist is to balance material properties in order to achieve, for example, a strong, corrosion resistant steel to endure Arctic conditions without being too brittle or expensive, or a nickel turbine blade for an aeroplane jet engine that can survive harsh high temperature conditions for tens of thousands of flight hours. Inevitably, some parts might fail and it is the job of the metallurgist to do failure analysis in controlled conditions to be able to reliably predict this and hence ensure it does not occur during use.

Richard Feynman (Nobel Prize winner for Physics) once asked the following question:
“If, in some cataclysm, all of scientific knowledge were to be destroyed, and only one sentence passed on to the next generations of creatures, what statement would contain the most information in the fewest words?”
His answer: ‘The atomic hypothesis - that all things are made of atoms’
Everything in the universe is made up of tiny building blocks called atoms. Atoms are incredibly small. If you were to line up atoms along the edge of a sheet of paper, there would be around a billion (1,000,000,000) of them! In a glass of water there are around 1,000,000,000,000,000,000,000,000 atoms – that is more than the number of stars in the universe!
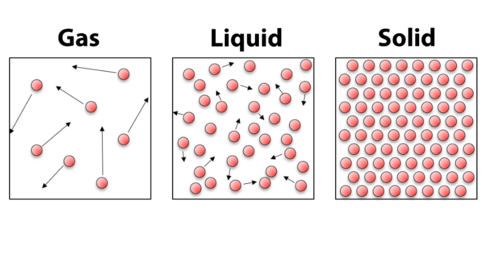
In a gas the atoms are quite spaced out and have lots of energy, and bounce around all over the place. It is this energy that means that gases expand to fill containers (like the room you’re sitting in!). In a liquid the atoms have less energy, but still move around bumping into each other. This arrangement of atoms means that liquids are able to flow and fill the bottom of containers. For a solid, the atoms are fixed in place – they may have a little bit of thermal energy causing them to shake or shiver, but on the whole they don’t move.
The arrangement of the atoms and the structures they form is therefore incredibly important to the properties of the material. We can change these structures by changing the atom type (i.e. changing the atom size and electronic properties). Different types of atoms are called elements, some of which are quite familiar – Oxygen, Carbon, Nitrogen, Iron, Gold. Chemists are interested in understanding how the different elements interact with one another and often use the periodic table (see Fig), which lists all the known elements.
In metals, atoms pack together closely, in specific arrangements, building the metal in layers. This can be seen in the diagram below. The drawing on the left show how the atoms fit together in the individual levels, and the picture on the right shows how the layers can stack on top of each other!
When the atoms are packed as tightly as possible the layers can slide over each other - imagine each of the layers in the diagram sliding over one another as you push or pull the structure.
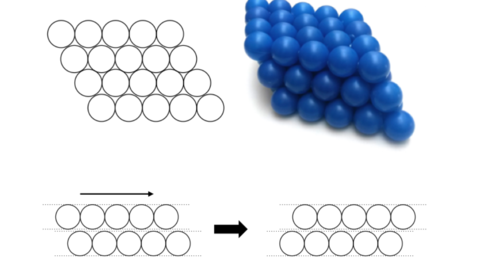
The strength and ductility (how easily it can be pulled into a wire) are due to this sliding. Whenever you deform a piece of metal, for example bending a paper clip of atoms, the planes of atoms are sliding over one another. The harder it is for the layers to slide, the stronger the metal is as it is harder to deform. So how can we make it harder for the layers to slide over one another
How can we use atoms to make them stronger?
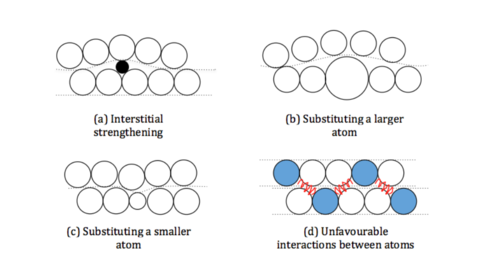
We must find a way to make it harder for the layers to slide over each other. There are four ways we can do this:
a) Very small atoms, such as carbon and boron can fit in between the layers becoming obstacles to the sliding atoms as the layer must distort to get round them. This is known as Interstitial Strengthening.
b) Replace atoms in the layers with slightly larger ones. The atoms surrounding this larger ones must distort to accommodate it, including those in the neighbouring layers. The layers are no longer flat and so, there is no longer a smooth path for the layers to slide past.
c) Replace atoms in the layers with slightly smaller ones. The surrounding atoms move in towards the smaller atoms distorting the layers. This results in the same effect as above. The layers are no longer straight to slide.
d) In some cases when element B prefers to be next to element A, resulting in an ordered structure where B is never next to B and A is never next to A. If the layers were to slide over each other the order would be upset and B would be next to B and A next to A. As this ruins the order it is very hard for the atoms to slide past each other.
When we design new materials we sometimes use all four to strengthen our metal. Just doing this we can make a metal which is so strong 15 cars can hang off it without it deforming. However, increasing the strength has downsides, we have now made it more brittle (this means when it breaks, it is more likely to snap - like a bar of chocolate!). There is a careful balance which means we must design a different alloy for each different requirement.
This fantastic video from our collaborators, the Naked Scientists, helps to explain exactly what an alloy is!
This link will take you to YouTube, which is an external webpage.
