Articles
Back in 2014, nearly 10 million people in the UK watched ‘The Great British Bake Off’ contestants make a baked Alaska, and the now infamous ‘Bin-gate’ scandal in which one contestant Ian, threw away his ice-cream after another contestant removed it from the fridge. But how many of them actually stopped to think about the science behind making a baked Alaska? Moreover, did they realise how it is the same science used in jet turbine engines to stop them from melting mid-flight?
Jet engine turbines operate at around 1800°C. To put that in context, lava flows are typically at a temperature of around 1100-1200°C. We therefore have a problem! The metals used in this part of the engine, known as superalloys, typically melt at around 1400°C. This is like putting an ice cube or icecream in a flame and expecting it not to melt. It’s not going to happen… So why doesn’t the engine turn into a puddle mid-flight?
Let’s go back to baked Alaska. You take a cake, put ice cream on top and then cover the whole thing in uncooked meringue. The whole thing is then put in a very hot oven, to quickly cook the meringue, without letting the ice-cream melt. But how? The answer is the meringue and the cake. Both the meringue and cake surround the icecream preventing the heat from getting to the icecream and preventing it from melting. Cake and meringue both contain lots of air pockets that act as a cushion insulating the ice-cream and preventing it from melting.
So what about the engine? Here the same idea is used. By coating the metal in a type of ceramic called a Thermal Barrier Coating (TBC) we prevent the blade from reaching the extreme temperature of the engine. The picture below shows a turbine blade from a real engine. You may notice lots of tiny holes all over the surface. ‘Cold’-air is constantly bled out of these holes, creating a cushion of air around the blade that insulates the metal, stopping it from experiencing the extreme temperatures of the engine.
The combination of these two effects, a protective coating and cooling cushion of air, ensure that the metal never reaches it's melting point and so the materials and the engine can operate in environments above their melting point. Using the simple principle of insulation we can therefore prevent jet-turbine engines from melting and also make a super yummy dessert.

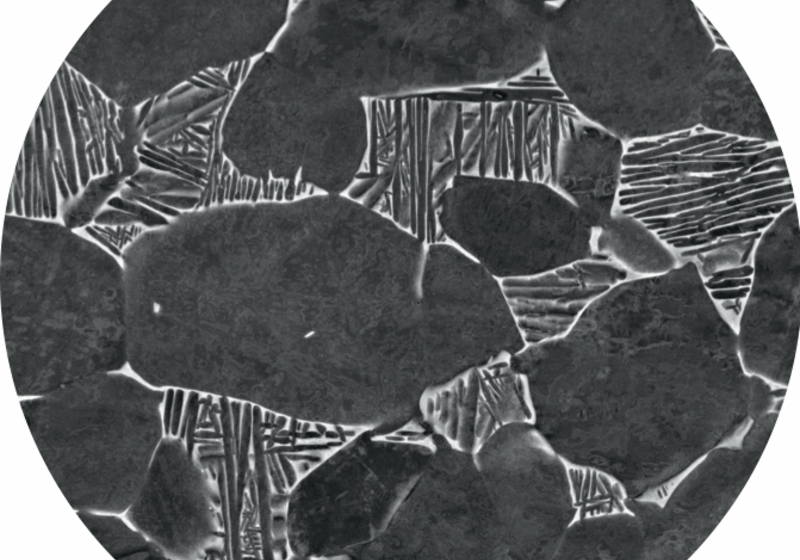
The simple answer is that they are all crystals! Most people when they think of crystals think of Diamond, Quartz, Rubies and other gemstones. However, there are many more crystals all around us in the world. What makes a crystal a crystal is a repeating structure. Take a look at the classic MC Escher drawing below.
If you look at the pattern you will notice that it is made up of the same unit repeated over and over again (shown by the red shape). A crystal is something made of a repeating structure, just like the Escher drawing shown. In the case of a real crystal this repetition occurs in three dimensions. Therefore anything that has this type of repeating structure is called a crystal. This includes diamonds, metals and chocolate.

The crystal structure of chocolate is what is responsible for the shiny nature of dark chocolate and for the crack you get when you break chocolate. If you try cheap or old chocolate then you’ll find that it is not as shiny and doesn’t snap properly, because the structure is less crystalline!
For us, as metallurgists, it is incredibly important to understand the crystal structure of the metal to understand the properties. A good analogy is with lego blocks. By changing the elements (in a process called alloying) that go into the material we can change the crystal structures. This is basically like making different sizes and shapes of lego blocks. By changing the size and shape of the blocks we are able to build different structures with different properties. Therefore, by changing the composition of the alloy we can make changes to the crystal structure and so to the properties of the overall material; allowing us to design new materials with more desirable properties.
You may, at some point it your life, have heard someone say something along the lines of:
Golly! It’s so cold today; it could freeze the balls off a brass monkey
Etymologists have found the earliest use of variants of this phrase as early as the 19th Century, but these generally refer to freezing the hair, tail, nose or ears off the poor monkey. However, urban legend suggests that the phrase actually comes from much earlier and is based on metallurgical principles!
For the 16th Century navy, cannons were the most important method of combat. In case of sudden attack by pirates or in naval battles, it was necessary to keep cannonballs close to the cannons. To prevent the balls from rolling around on the deck a brass metal frame, known as a ‘monkey’, was used. By stabilising a layer of cannonballs on the bass of the monkey, the next layer of cannonballs simply sites in the holes made by the previous layer. Therefore, if you start with a square or triangular arrangement you can build up the structure into a pyramid. You can see this in the picture below:

This problem, of how to stack cannonballs, was first discussed by famous scientist Johannes Kepler in 1611, who showed that a triangular based pyramid was the most efficient way of stacking cannonballs. Not only is this interesting if you’re trying to stack cannonballs, but it turns out that this stacking actually reflects the way that many metals are structured. Atoms in a metal behave just like the cannonballs and stack one layer on top of another, sitting in the holes made by the previous layer. By understanding the way the atoms in a metal stack we can begin to understand some of their properties and so design materials with more desirable properties!
Returning to the monkey - in order to stabilise the cannonball structure, you just need to stablise the base layer. However, when it gets cold, metals are observed to contract. This would destabilise the base layer and lead to the cannonball structure collapsing, causing the balls to come loose and roll around the deck.
So why does the metal contract as it gets cold? As we’ve discussed metal atoms stack together in the structure, sitting in fixed positions. Even though they sit in specific positions in the structure, the atoms are always shaking and vibrating, due to thermal energy. As the temperature drops the atoms in the structure vibrate less, allowing them to squeeze closer together, causing the entire metal to contract slightly. In the same way if you heat the metal it will expand.
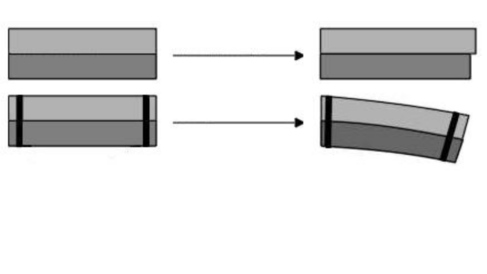
This can be really useful in some cases such as bimetallic strips. Different metals contract and expand at different rates. Therefore, if we strap together two different metals and heat them, they will expand by different amounts. The only way for the metal to cope is for it to bend. We can therefore use this to make components that are sensitive to temperature, for example thermostats!
Tests done recently have suggested that in fact the metal would not contract enough on a ‘brass monkey’ to cause the cannonball tower to collapse, suggesting that this is just an urban legend. Even if it’s not true, it’s a pretty good story and demonstrates some interesting metallurgical principles!
Nestled in the Oxfordshire countryside is the Rutherford Appleton laboratory, home to the Diamond Light Source and the ISIS Neutron and Muon source. The two facilities are large scale particle accelerators and by using them scientists are able to understand fundamental science occurring in a broad range of fields; from the structure of biological proteins all the way to deformation in aviation parts. But how do these particle accelerates help us understand these materials?

In order to look at something small, like an insect, you might use a magnifying glass. For something even smaller like bacteria, you might require a optical microscope. These work by focussing the light reflected off an object to form an image that we can see. Even smaller, and you’ll need an electron microscope. These use electrons rather than light to form the images, which leads to much greater magnification. Unfortunately, it turns out that these sort of image forming microscopes have a limit, and if we want to look at something as small as an atom, we need to use an alternative method instead. The answer comes from a principle known as diffraction.
Have you ever stood on a beach and watched what happens when a water wave goes through a gap, like a sea-wall? As it passes through the sea wall, the waves spreads out in a circle (see image). This process is called diffraction and is a unique property of waves.

So what happens if you have two gaps in your wall? Well the same thing, the water passes through both holes and then spreads out on the other side. But now there are two waves that are moving towards each other (one from one whole and one from the other). We can guess what will happen. If the high point in one wave meets a high point in the next wave, then the waves add together to make a much large wave. If a high point in one meets a low point in the other, then they cancel out. This is known as interference, either constructive or destructive, and results in a distinct pattern, called a diffraction pattern, of high and low waves; or in the case of light diffracting a series of light and dark lines.
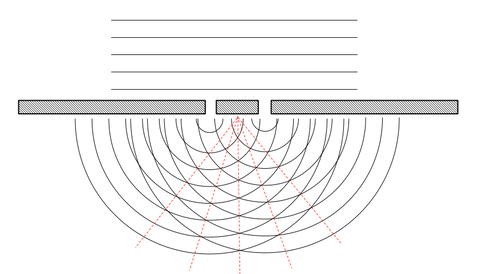
But what has this got to do with the structure of matter? Nearly 100 years ago scientists realised the the gaps between layers of atoms in a crystal could act just like the gaps in a sea wall and could produce a diffraction pattern. By taking light and passing it through the crystal and recording a diffraction, we can then calculate the spacing and positions of atoms in the structure. It was found that the specific light that was needed were X-rays, and they needed to be incredibly bright. Synchrotrons, like the Diamond Light Source, accelerate particles as a way of creating very high energy X-rays that are brighter that the sun. It was also found that we can use very fast moving particles called Neutrons in a similar way to the light to probe the structure of materials. By understanding the structure of the materials, using these facilities, we can begin to identify and explain the link between structure and properties. We can examine how the material changes as we heat it, put it under stress, break it etc. and use this knowledge to design better materials for the future.
In order to understand the properties of the metal we need to examine it's structures on all length scales, right down to the atomic. To do this we use powerful microscopes known as electron microscopes to look at the metal up close. On the Engineering Atoms stand we have an example of a Scanning Electron Microscope that you can use yourself to look different metals up close. But how do these microscopes work? Below is a schematic showing what is taking place inside the microscope:
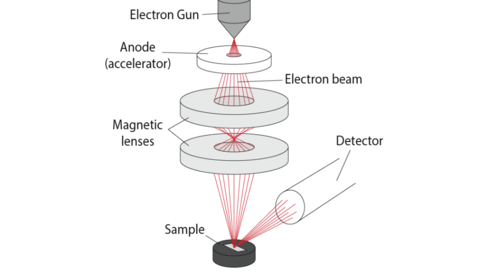
Electron Gun: Electrons are produced by either heating a filament, or by applying a strong electric field to it. Filaments are often made of tungsten, and have a very sharp tip so that a very narrow stream of electrons is emitted.
Anode: The electrons are accelerated down the column of the microscope by the anode. The anode is negatively charged so repels the negatively charged electrons, forcing them to accelerate.
Magnetic lenses: The electrons are focused onto the sample by magnetic lenses. These ensure a very narrow beam of electrons hits the sample.
Sample: The sample scatters the electrons. The amount of scattering (and the number detected at the detector) can depend on many factors, including: sample height, sample chemistry, and sample crystal structure. X-rays are also produced by the electrons hitting the atoms in the sample. We can analyse these X-rays to tell us which elements exist in the sample.
Detector: The beam is scanned across the sample, and the signal from every point detected. There are many different types of detector that can detect electrons with different energies travelling in different directions. Using different detectors gives us different information about our sample.